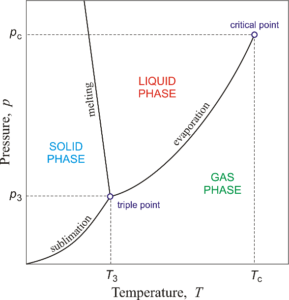
A phase diagram is a chart that represents the equilibrium exists between different phases that coexist in the same closed system. There are three major phases that any substance can exist: solid phase, liquid phase and gas phase. Summary – Phase Diagram vs Equilibrium DiagramĪ phase is a homogeneous portion of a system that has uniform chemical and physical properties. Both terms refer to the chart that summarizes the details on conditions of thermodynamically distinct systems that coexist in equilibrium with each other.
Binary Phase DiagramsĪ binary phase diagram shows the equilibrium between two substances that exist in the same system. These diagrams show the three phases of a substance separated from equilibrium boundaries (such as in Figure 1). These are the simplest form of phase diagrams. Sublimation is the conversion of a solid into gas phase directly, without going through a liquid phase. Vaporization is the conversion of a liquid into vapor phase (gas phase) whereas condensation is the conversion of a vapor into liquid. Here, fusion is the melting or freezing that involves the phase transition between gas phase and liquid phase.

There is no difference between phase diagram and equilibrium diagram. There are three major phases that a substance can exist: solid, liquid and gas phase. A phase is a homogeneous portion of a system that has uniform chemical and physical properties. These diagrams give details on conditions of thermodynamically distinct phases that coexist in equilibrium with each other. Phase diagrams or equilibrium diagrams are charts or graphs showing the relationship between temperature, pressure and the composition of any system. Key Difference – Phase Diagram vs Equilibrium Diagram
